Voltage-gated sodium (Nav) channels are pivotal transmembrane proteins that mediate the initiation and transmission of bioelectrical signals. They are involved in key physiological processes, including neurotransmitter release, muscle contraction, hormone secretion, and sensory perception. Functioning as "molecular gates" on the cell membrane, the voltage-sensing domains (VSDs) of these channels detect changes in membrane potential. In response to electrical field, the VSD undergoes a conformational rearrangement that triggers pore opening, allowing sodium ions to flow inward and initiate an electrical signal. Notably, the open state persists only milliseconds before the channel rapidly inactivates, a process known as "fast inactivation". This property is believed to ensure the "all-or-none" nature and unidirectional propagation of action potentials, providing precise spatiotemporal control for nerve impulses and muscle contraction. However, owing to its transient nature, “capturing” a high-resolution structure of the open state remains a big challenge and a major technical bottleneck in this field. [1–3]
On November 18, 2025 (Beijing Standard Time), a team led by Nieng Yan, Jian Huang, and Xiao Fan from the Shenzhen Medical Academy of Research and Translation, in collaboration with Chen Song’s team from Peking University, posted a preprint on LangTaoSha Preprint Server, entitled "Open-state structure of veratridine-activated human Nav1.7 reveals the molecular choreography of fast inactivation".
This study reports the first high-resolution three-dimensional structure of the open-state human Nav1.7, offering key evidence for understanding the molecular mechanism of fast inactivation. To overcome the challenge of stabilizing this transient state, the team treated the channel with veratridine, a compound that acts as a potent Nav opener. Using cryogenic electron microscopy, they successfully reconstructed the high-resolution three-dimensional structure, thereby visualizing the core "open" conformation within the channel's functional cycle.

Fig. 1 LangTaoSha preprint
在本研究中,研究团队成功捕获两种与藜芦定结合的Nav1.7复合物结构(图2)。其中一种构象中藜芦定嵌入IFM结合角区域(位点I),与其他常见失活态结构类似;而在另一种构象中,藜芦定贯穿中央空腔(位点C),该构象显示胞侧门控收缩处的孔径达8.2埃,超过水合钠离子的直径(图3)。分子动力学模拟显示,该构象所对应的通道完全通透,并在模拟中呈现与实验结果相近的电导值,代表处于激活开放态的功能性结构(图3)。通过对比开放态与失活态的精细结构差异,研究团队得以清晰描绘Nav1.7由开放向失活转变的构象变化路径,为理解其门控及快速失活的分子机制提供了直观的结构基础(图4)。
In this study, the team successfully obtained two veratridine-bound Nav1.7 complexes (Fig. 2). One, with veratridine inserted in the IFM-binding corner (site I), resembles other inactivated structures. The other, wherein veratridine transverses the central cavity (site C), exhibits a constriction diameter of 8.2 Å at the intracellular gate, which exceeds the diameter of a hydrated sodium ion (Fig. 3). Molecular dynamics simulation analysis indicates that this conformation corresponds to a fully conductive pore, yielding conductance comparable to the experimental value, thereby representing a functional activated open state (Fig. 3). A high-resolution structural comparison between pore opening and subsequent fast inactivation provides an intuitive structural basis for understanding the molecular mechanisms of channel gating and fast inactivation (Fig. 4).
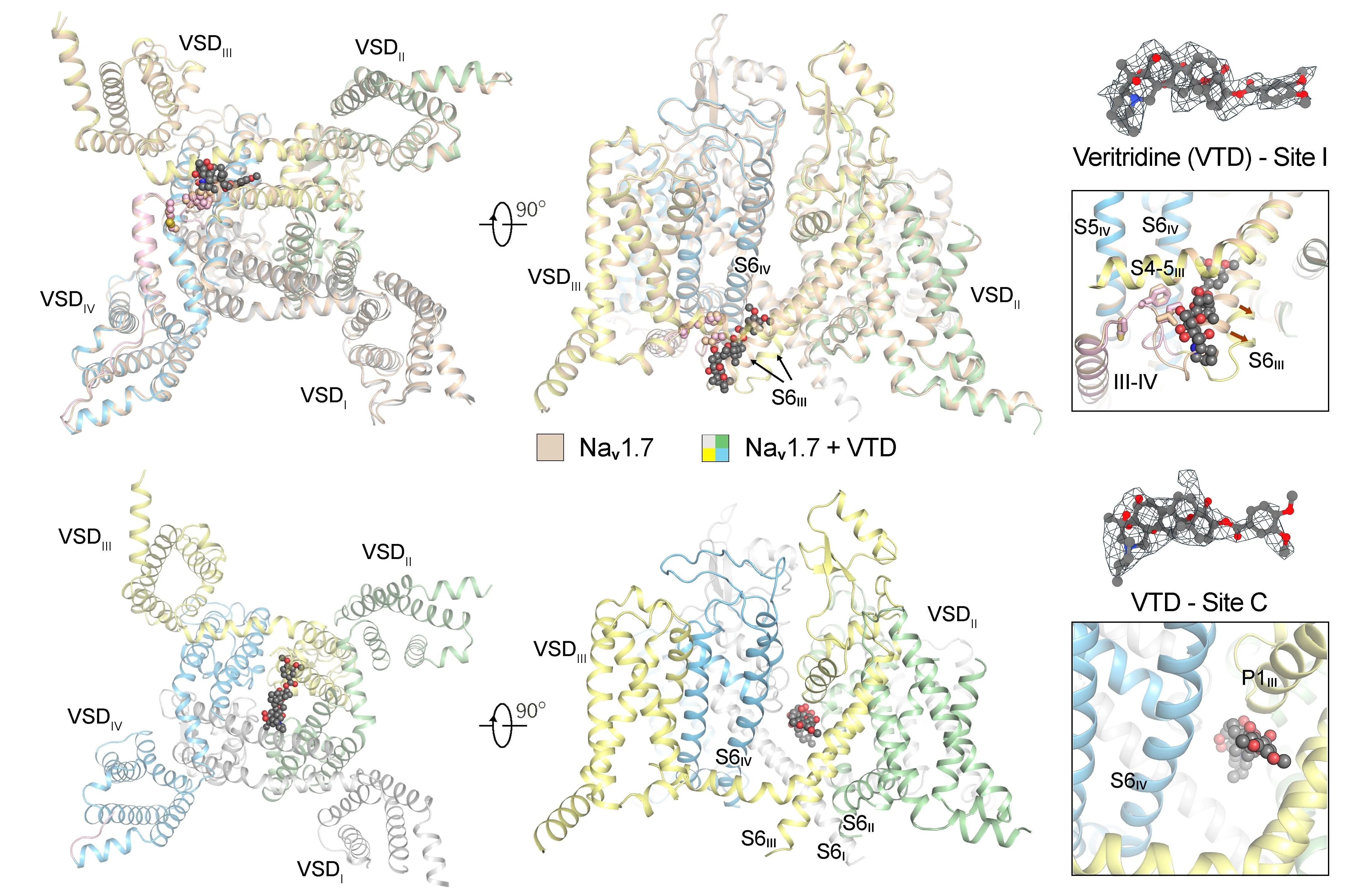
Fig. 2 Two distinct binding modes of veratridine to Nav1.7
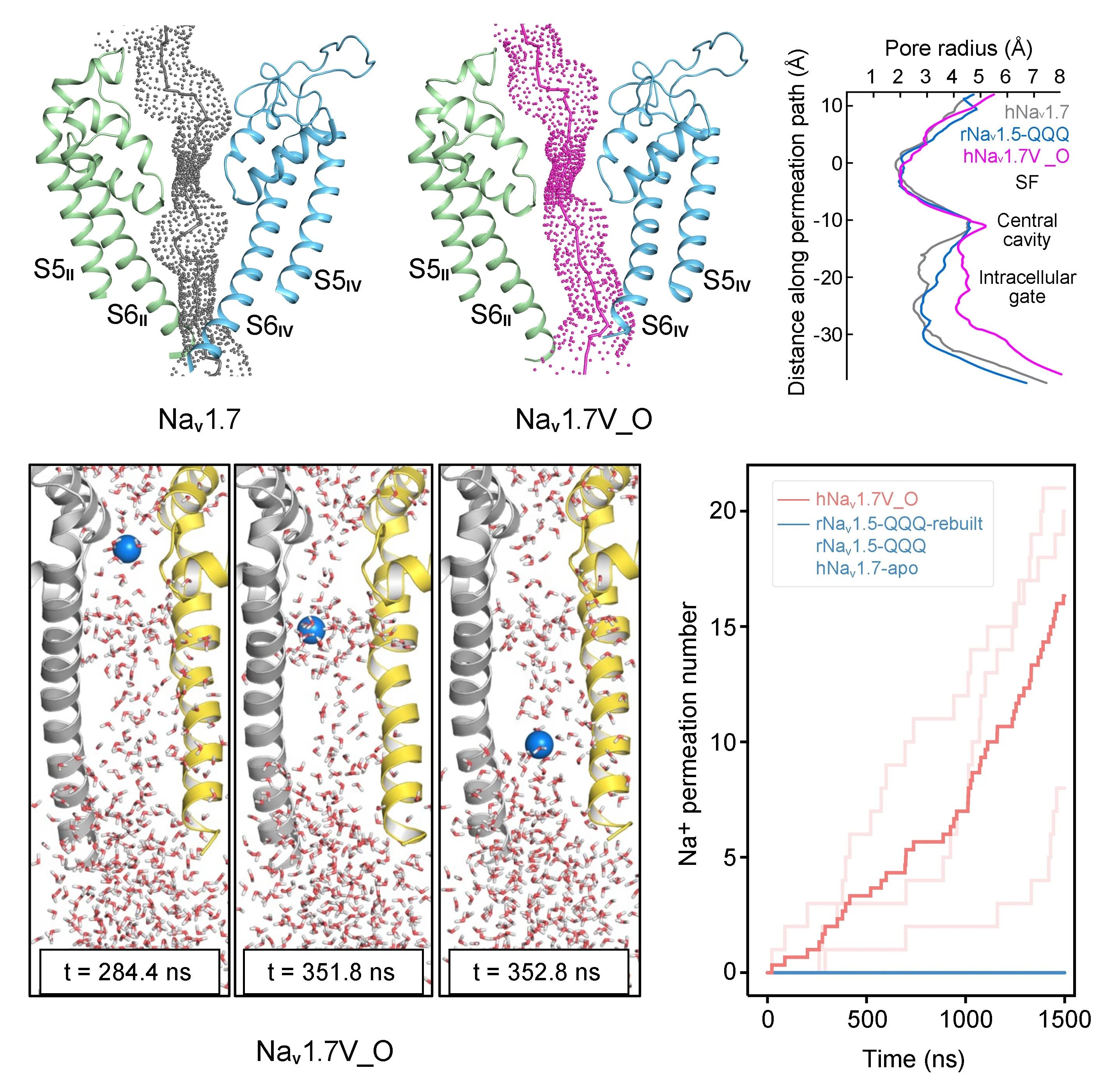
Fig. 3 The open-state Nav1.7 is fully conductive in molecular dynamics simulations
Dysfunction of Nav channels is associated with a variety of serious diseases, including epilepsy, cardiac arrhythmia, chronic pain, and congenital insensitivity to pain. Over a thousand disease-associated mutations have been identified across nine human sodium channel proteins. Moreover, these channels are direct targets for several classes of clinically important drugs, such as local anesthetics, antiepileptics, and antiarrhythmics, which typically exert their therapeutic effects by selectively binding to specific channel states, such as the open or inactivated state. [4-7]
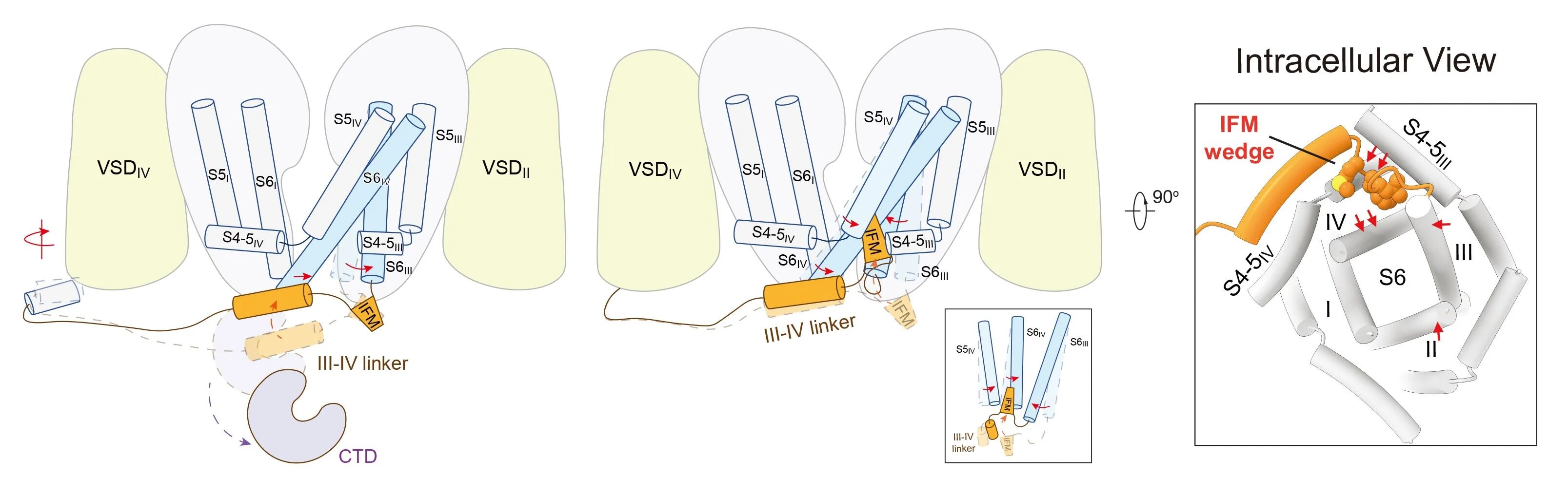
Fig. 4 Molecular mechanism model for fast inactivation of sodium channels
Therefore, obtaining a high-resolution open-state structure of sodium channels such as Nav1.7 achieves three key advances: it enables a systematic, atomic-level elucidation of channel gating principles and the mechanism of fast inactivation; it provides a solid structural basis for interpreting the functional defects caused by numerous pathogenic mutations; and, most importantly, it offers an unprecedented structural template for designing and optimizing next-generation state-selective sodium channel modulators with improved selectivity and reduced side effects. This breakthrough holds significant theoretical and practical value for drug discovery targeting neurological, cardiovascular, and chronic pain disorders.
This study was led by co-corresponding authors Nieng Yan (Founding Dean, Shenzhen Medical Academy of Research and Translation; Director, Shenzhen Bay Laboratory), Jian Huang (Junior Principal Investigator, Shenzhen Medical Academy of Research and Translation), Xiao Fan (Junior Principal Investigator, Shenzhen Medical Academy of Research and Translation), and Chen Song (Researcher, Center for Quantitative Biology, Peking University). Xiao Fan (Junior Principal Investigator, Shenzhen Medical Academy of Research and Translation), Jiaofeng Chen (PhD, School of Life Sciences, Tsinghua University), and Lingfeng Xue (PhD, Center for Quantitative Biology, Peking University) are the co-first authors. Other contributors include Huan Wang (PhD), Tong Wu (PhD), and Xueqin Jin (Research Associate) from School of Life Sciences at Tsinghua University, as well as Xiaoshuang Huang (Research Associate) and Fangzhou Lu (PhD Candidate) from Shenzhen Medical Academy of Research and Translation. This work was funded by the Shenzhen Medical Academy of Research and Translation, National Natural Science Foundation of China, and Beijing Frontier Research Center for Biological Structure.
References:
[1] Hille, B. Ion channels of excitable membranes, 3rd edition. (2001)
[2] Hodgkin, A. L., & Huxley, A. F. (1945). Resting and action potentials in single nerve fibres. The Journal of Physiology, 104(2), 176.
[3] Huang, J., Pan, X., & Yan, N. (2024). Structural biology and molecular pharmacology of voltage-gated ion channels. Nature Reviews Molecular Cell Biology, 25(11), 904-925.
[4] Woolf, C. J., & Mannion, R. J. (1999). Neuropathic pain: aetiology, symptoms, mechanisms, and management. The Lancet, 353(9168), 1959-1964.
[5] Catterall, W. A. (2014). Sodium channels, inherited epilepsy, and antiepileptic drugs. Annual Review of Pharmacology and Toxicology, 54(1), 317-338.
[6] Huang, W., Liu, M., Yan, S. F., & Yan, N. (2017). Structure-based assessment of disease-related mutations in human voltage-gated sodium channels. Protein & Cell, 8(6), 401-438.
[7] Rubinstein, M., Patowary, A., Stanaway, I. B., McCord, E., Nesbitt, R. R., Archer, M., ... & Brkanac, Z. (2018). Association of rare missense variants in the second intracellular loop of NaV1. 7 sodium channels with familial autism. Molecular Psychiatry, 23(2), 231-239.
Subscription successful! Thank you for following SMART.