Cryo-electron tomography (Cryo-ET) enables three-dimensional reconstruction of biological macromolecules within cells and tissues from sub-nanometer to near-atomic resolution, preserving their native state by collecting biological simples at low temperature from multiple tilt angles. This technique integrates structural information across molecular, cellular, and tissue levels, making it a pivotal tool for in situ structural biology and the discovery of novel cellular phenomena. While Cryo-ET is well-established for single-cell analysis, its application to more physiologically relevant multicellular tissues remains technically challenging. A major bottleneck is cryo-focused ion beam (Cryo-FIB) milling, a critical step for preparing tissue lamellae suitable for Cryo-ET data collection and a rate-limiting step for in situ structural studies. However, current Cryo-FIB method requires improvement in operational difficulty, lamellae stability, and preparation efficiency.
Meijing Li's team from Shenzhen Medical Academy of Research and Translation has recently published two back-to-back papers in Biophysics Reports, entitled "Step-by-step procedure for an optimized serial lift-out cryo-focused ion beam milling technique in tissue analysis" and "Structural plasticity of mouse intestinal microvilli revealed by tissue-level cryo-ET".
The team developed and systematized an optimized serial-lift-out cryo-FIB workflow for multicellular tissue samples. This approach significantly enhances lamellae stability, preparation efficiency, and experimental repeatability, while reducing dependence on operator experience. Core optimizations include a six-sided attachment scheme and an adjustable slit design, which enable efficient extraction and precise mounting of tissue blocks of varying sizes onto pre-milled electron microscopy grids. This workflow combines high-pressure freezing and cryo-correlative light to electron microscopy (Cryo-CLEM), establishing a complete, generalized pipeline from cryofixation to final lamella preparation for in situ structural studies at the tissue level. This method is applicable for in situ structural studies of specific cells, subcellular structures, or macromolecules within diverse biological tissues.
Using the optimized serial-lift-out cryo-FIB and Cryo-ET workflow, Meijing Li’s team obtained extensive ultrastructural data from mouse intestinal tissues. Analysis revealed a variety of branched microvilli, with their proportion significantly higher than that reported in nematode intestines. This finding indicates a previously underappreciated structural plasticity in the mammalian brush border and provides direct structural evidence supporting the hypothesis that "a fission-like mechanism may exist in microvilli renewal".

Link to full text: https://www.biophysics-reports.org/article/doi/10.52601/bpr.2025.250039

Link to full text: https://www.biophysics-reports.org/article/doi/10.52601/bpr.2025.250038
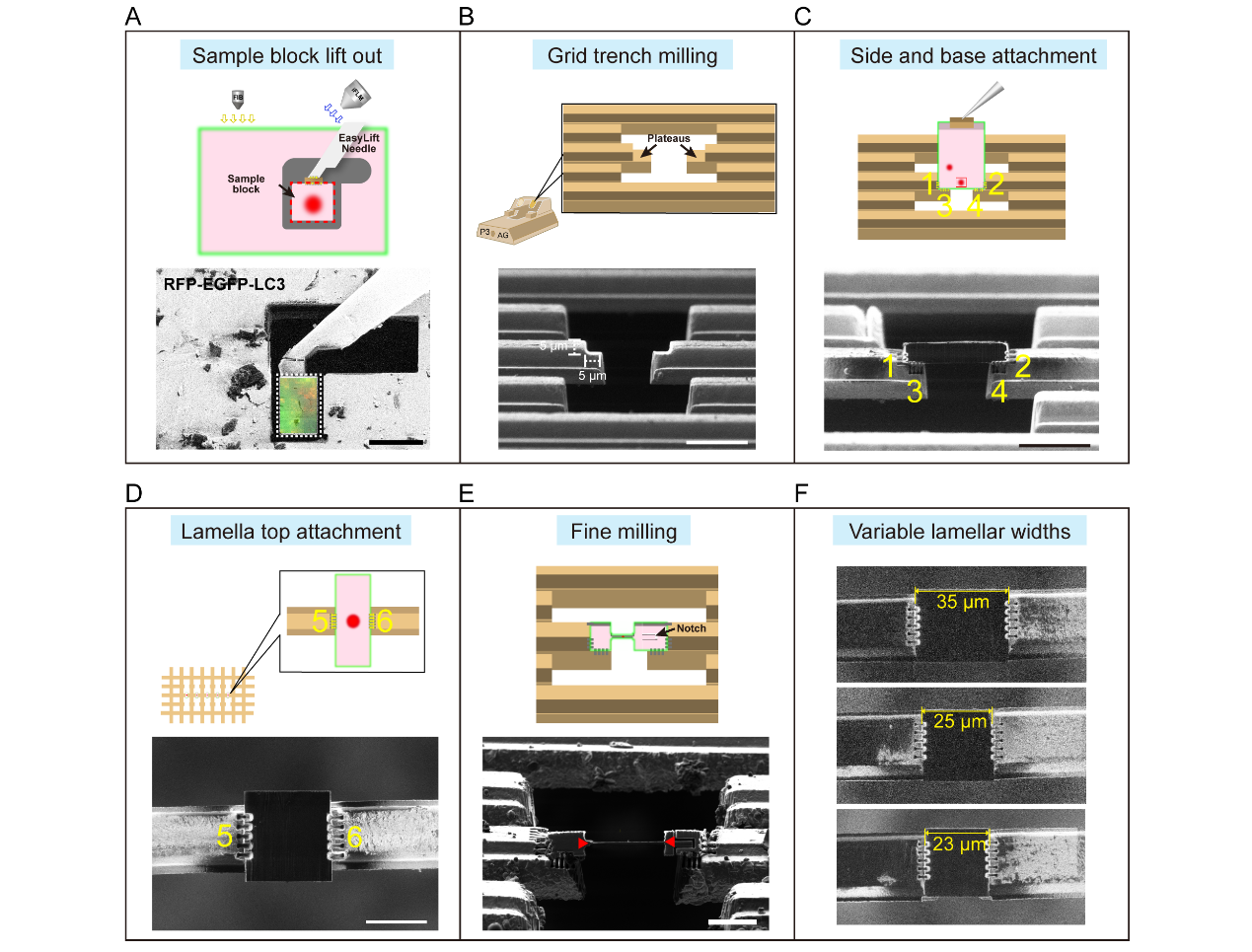
Fig. 1 Optimized serial lift-out cryo-FIB workflow
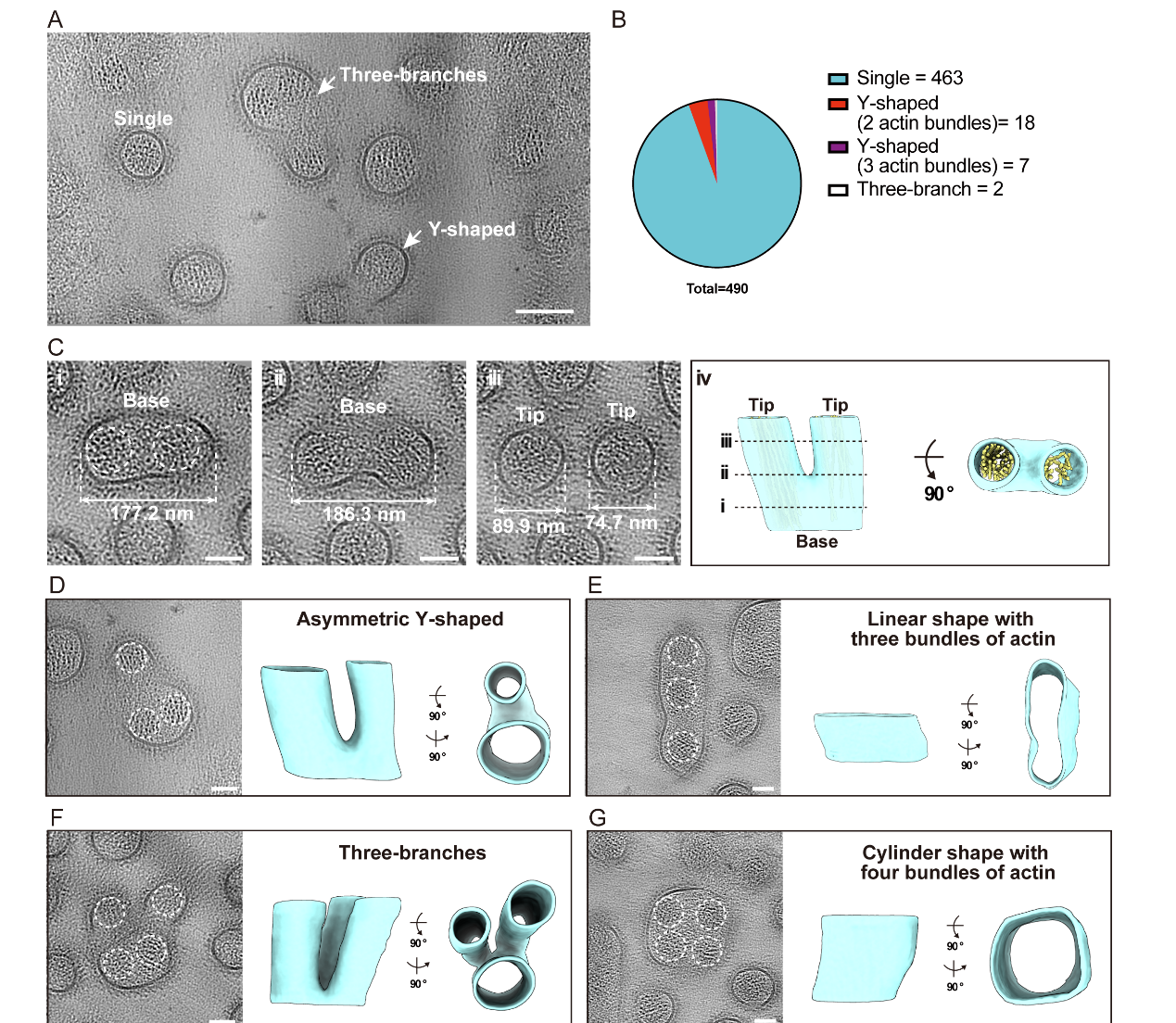
Fig. 2 In situ structure of various branched microvilli in mouse intestine
Meijing Li (Researcher, Institute of Bio-Architecture and Bio-Interactions, Shenzhen Medical Academy of Research and Translation) is the correspondent author for both papers. Jiaming Liu (Research Associate) and Xiaoyu Tang (PhD Candidate) from Institute of Bio-Architecture and Bio-Interactions at Shenzhen Medical Academy of Research and Translation are the first authors of the respective studies. Other contributors include Huanhuan Huang (PhD Candidate), Jingtao Zheng (PhD Candidate), Zihang Yu (PhD Candidate), and Peng Wang (Postdoctoral Fellow) from Institute of Bio-Architecture and Bio-Interactions at Shenzhen Medical Academy of Research and Translation. The biological structure analysis platform and high-performance computing platform of Shenzhen Medical Academy of Research and Translation provided electron microscopy data collection and computational support. This work was funded by Shenzhen Medical Academy of Research and Translation and National Key Research and Development Program of China.
Subscription successful! Thank you for following SMART.