Voltage-gated sodium channels (Nav) are core transmembrane proteins that mediate bioelectric signals, widely participating in key physiological processes such as neurotransmitter release, muscle contraction, hormone secretion, and the perception of external stimuli. Navdysfunction can lead to diseases such as epilepsy, arrhythmias, and chronic pain. Acting as "electric gates" on the cell membrane, Nav channels sense changes in membrane potential through the voltage-sensing domain (VSD) and undergo conformational changes driven by electric field forces to open the gate. This allows sodium ion influx, which in turn triggers and propagates electrical signals. After opening, the channels remain open for only a few milliseconds before closing rapidly. This "fast inactivation" mechanism is considered the molecular basis for ensuring the "all-or-none" property and unidirectional conduction of action potentials, providing precise spatiotemporal regulation for processes such as nerve impulses and muscle contractions. Hence, the fleeting nature of the open state makes its structure difficult to capture.
On January 25, 2026 (Beijing Time), the teams of Nieng Yan, Jian Huang, and Xiao Fan from the Shenzhen Medical Academy of Research and Translation (SMART), in collaboration with Chen Song's team from Peking University, published an article titled "Open-state structure of veratridine-activated human Nav1.7 reveals the molecular choreography of fast inactivation" in the inaugural issue of Vita.

Using Cryogenic electron microscopy (cryo-EM), the article resolved thethree-dimensional structure of the human voltage-gated sodium channel Nav1.7 in its open state for the first time, successfully capturing the critical “molecular instant” of channel opening, and filling a key gap in revealing the core mechanism of sodium channel fast inactivation. In this study, they ingeniously introduced Veratridine, a lipid-soluble neurotoxin derived from Liliaceae plants, as a sodium channel agonist, and successfully captured two complex structures of Nav1.7 bound to Veratridine. In one conformation, Veratridine is embedded in the IFM-binding corner region (Site I), similar to previously reported inactivated state structures; in the other conformation, Veratridine penetrates the central cavity (Site C), presenting an activated state conformation. In this conformation, the pore diameter at the intracellular gating constriction reaches 8.2 Å, large enough to accommodate a hydrated Na+ ion of 7.2 Å for permeation. Further molecular dynamics simulation results indicate that the channel corresponding to this conformation is fully permeable, and the conductance values presented in the simulations are highly consistent with experimental results, confirming that this functional structure is indeed in an activated open state.
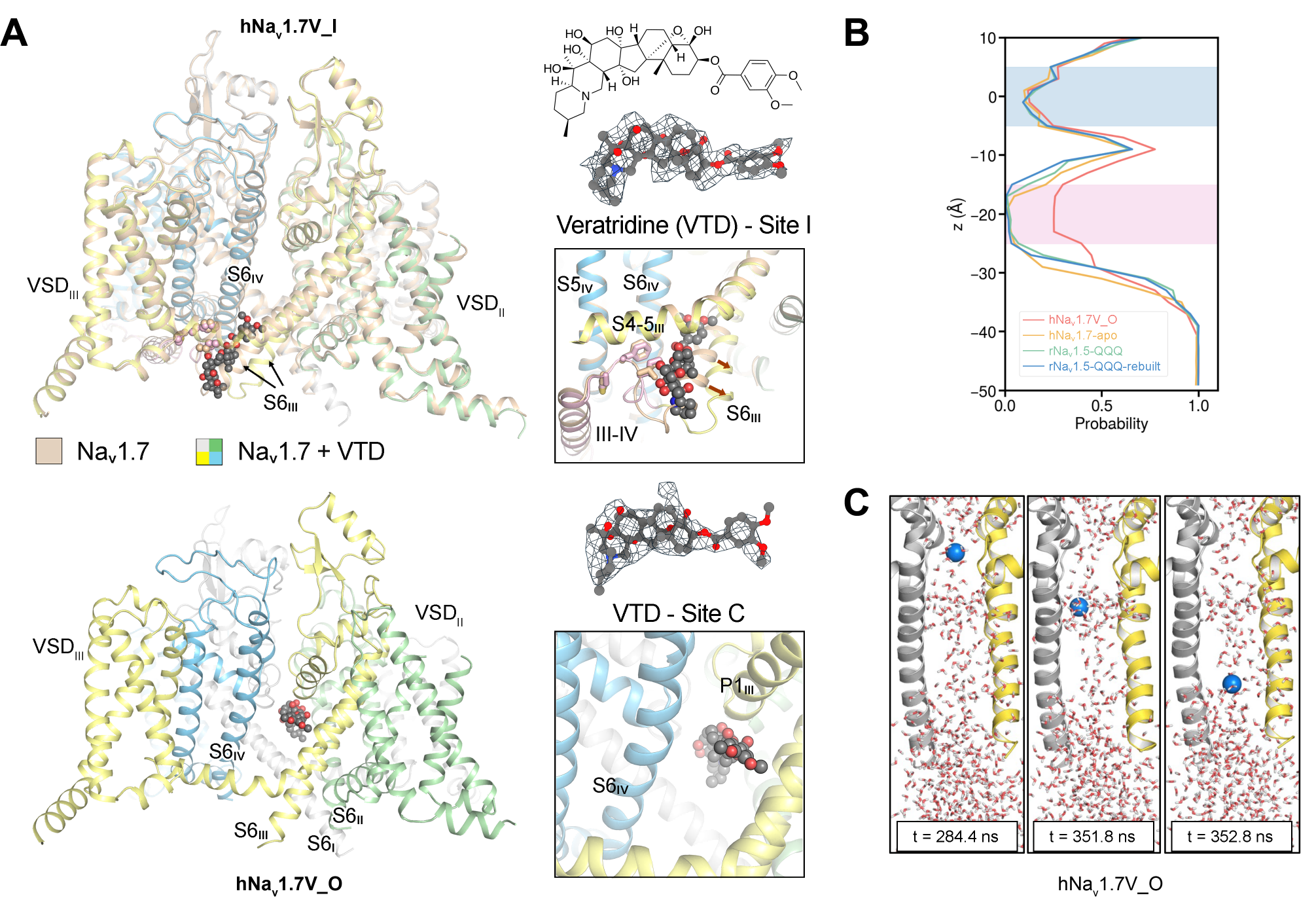
Two binding modes of Veratridine with Nav1.7 and molecular simulation experiment results
Based on this core structure, the research team conducted a systematic comparison with previously published structures of sodium channels in the inactivated state, clearly outlining the conformational change trajectory of Nav1.7 from opening state to inactivation. This provides a direct structural perspective for resolving the gating mechanism and fast inactivation process of sodium channels at an atomic level.
Sodium channels are important targets for clinical drugs such as local anesthetics, antiepileptic drugs, and analgesics. The high-resolution structural resolution of the open state of Nav1.7 will help deepen the understanding of its gating mechanism and fast inactivation process, providing a structural basis for explaining pathogenic mutations. More importantly, this structure provides a precise template for designing a new generation of sodium channel drugs that target specific functional states with high selectivity and low side effects. This holds significant application value for drug development for major diseases such as neurological disorders, cardiovascular diseases, and chronic pain.
Notably, this research was first published on the LTS Preprint Server (LTSpreprints.org) on November 18, 2025, becoming the first paper since the server launch. To date, it has accumulated nearly 11,000 views and over 3,000 full-text downloads. Subsequently, after rigorous peer review, the paper was officially published in Vita, a high-level international journal in the field of life sciences and biomedicine jointly established by the Open Life Science Alliance, becoming one of the important achievements published in the journal's inaugural issue.
This complete dissemination chain, from rapid public release on the preprint platform to official publication in a top journal, vividly reflects the publishing philosophy of the Vita journal: "zero OA fees, returning to the essence of academia." Furthermore, through the LTS Preprint Server, it realized the immediate open sharing of scientific research results, perfectly practicing the innovative dissemination model of "free instant preview across the entire network + rigorous peer review + journal publication." While balancing dissemination efficiency and academic credibility, this model effectively enhances the visibility and impact of research findings, fully demonstrating the unique advantage of an "independent platform + top international journal" collaborating to build an open academic ecosystem.

The achievement was previously published on the LTS Preprint Server (LTSpreprints.org)
Translation: Yang Shen
Proofreading: Fangzhou Lu
Subscription successful! Thank you for following SMART.