On December 17, 2025 (Beijing Time), the team led by Jian Huang and Xiao Fan at the Shenzhen Medical Academy of Research and Translation (SMART) published their latest research progress titled "Structural basis for the assembly and modulation of human M-channels" on the LTS Preprint Server (Figure 1). This study obtained the high-resolution cryo-electron microscopy (cryo-EM) structure of the human M-channel, systematically revealing its heteromeric assembly mode, voltage-sensing mechanism, drug recognition pattern, and a novel PIP2-dependent stepwise gating mechanism.
Concurrently, two other research papers on "M-channels" were also published on the LTS Preprint Server: a joint study by Huaizong Shen's team from Westlake University and Huaiyu Yang's team from East China Normal University, titled "Structural basis for heteromeric assembly and subthreshold activation of human M-channel"; and a joint study by Jin Zhang's team from Nanchang University and Panpan Hou's team from Macau University of Science and Technology, titled "Structural basis of the neuronal M-current by an asymmetric KCNQ2/3 channel assembly". These three independent studies complement and corroborate each other, jointly expanding our biological understanding of M-channels.

Fig.1 The research findings published on the Langtaosha preprint platform
Voltage-gated potassium (Kv) channels are essential "electrically sensitive valves" on the cell membrane. After cells such as neurons or cardiomyocytes depolarize, these channels are responsible for helping the membrane potential rapidly repolarize to a "resting" state. The precise regulation involving these channels ensures the accurate transmission of neural signals and the rhythm of heartbeats. Once the function of such channels becomes abnormal, it may trigger a variety of diseases such as epilepsy, cardiac arrhythmias, or neuropathic pain [1]. In addition to homomeric complexes, many Kv channels can also form heteromeric complexes, enabling the fine-tuning of voltage dependence, gating behavior, drug sensitivity, and susceptibility to disease mutations [2].
In the central nervous system, the KCNQ (also known as Kv7) family of channels acts as a "voltage stabilizer", preventing excessive excitation or disordered firing of neurons. Among them, the M-channel, primarily co-assembled by KCNQ2 and KCNQ3 subunits, serves as a "molecular brake" against neuronal hyperexcitability. Clinical studies have shown that a loss-of-function mutation in either subunit can induce epilepsy, making the M-channel a crucial target for antiepileptic drugs.
However, a long-standing key question remains: almost all known structural studies of KCNQ channels are based on homomeric complexes. How exactly do physiologically functional heteromeric M-channels assemble? How do different subunits achieve a functional asymmetry and coordination? The lack of direct structural evidence for these questions has long limited the field's in-depth understanding of their working mechanisms and drug modulation modes.
By co-expressing KCNQ2 and KCNQ3 proteins, Jian and Xiao's team successfully reconstituted functional heteromeric M-channels and confirmed their typical robust macroscopic currents using whole-cell patch-clamp techniques. Subsequently, by cryo-EM single particle analysis, the structures of the heteromeric channels were resolved: the predominant assembly ratio is three KCNQ2 to one KCNQ3 (3:1), while a minor 2:2 assembly stoichiometry also exists. In the 2:2 assembly, identical subunits are distributed diagonally, indicating a highly ordered assembly arrangement (Figure 2).
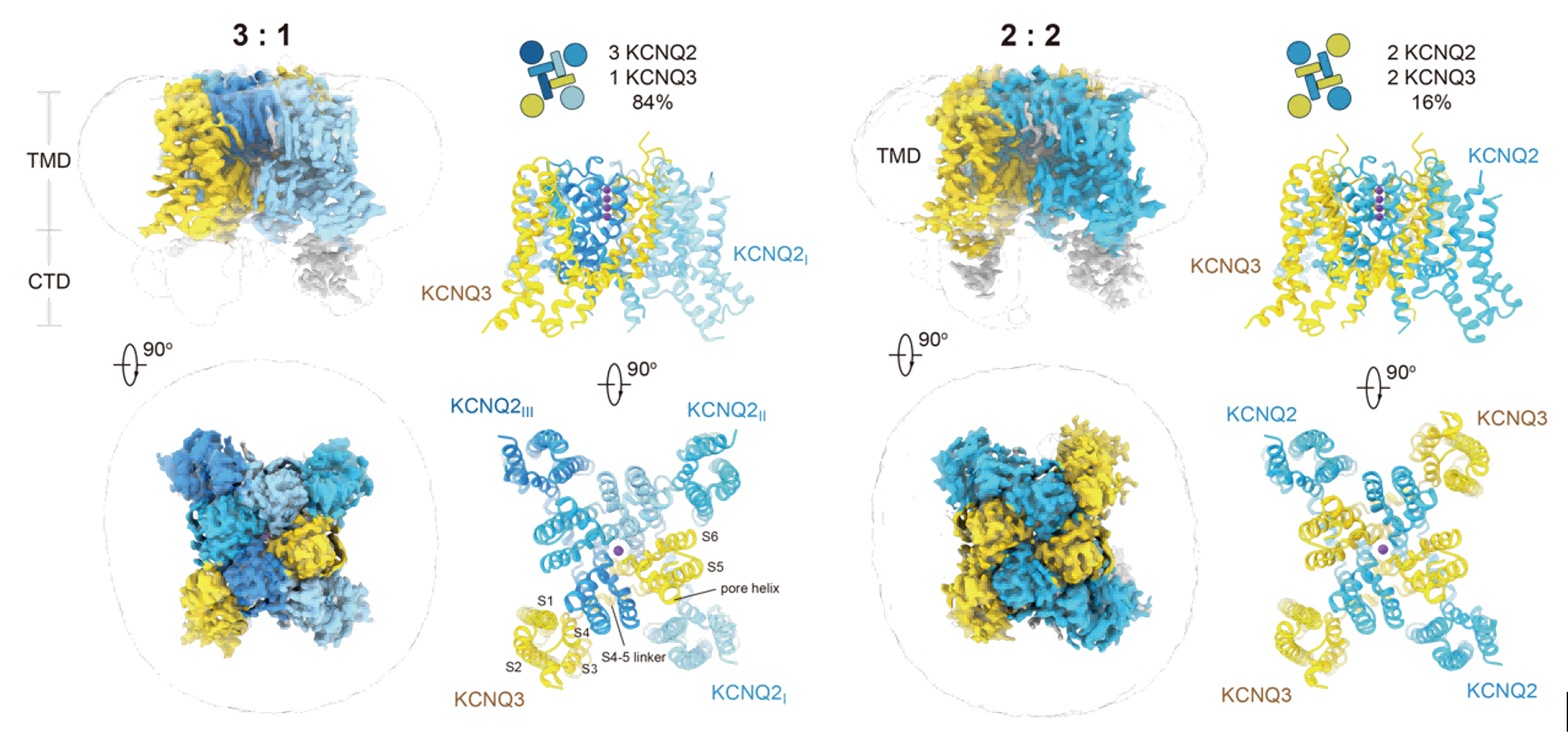
Fig.2 Heteromeric assembly mode of the M-channel
Further analysis revealed that compared to KCNQ2, the voltage-sensing domain (VSD) of KCNQ3 exhibits a pre-activated conformation. Functional experiments further demonstrated that when the VSDs of the M-channel are completely replaced with those of KCNQ3, the channel activates at more negative voltages, directly proving that KCNQ3 is the core element driving the low-threshold activation of M-channels (Figure 3).
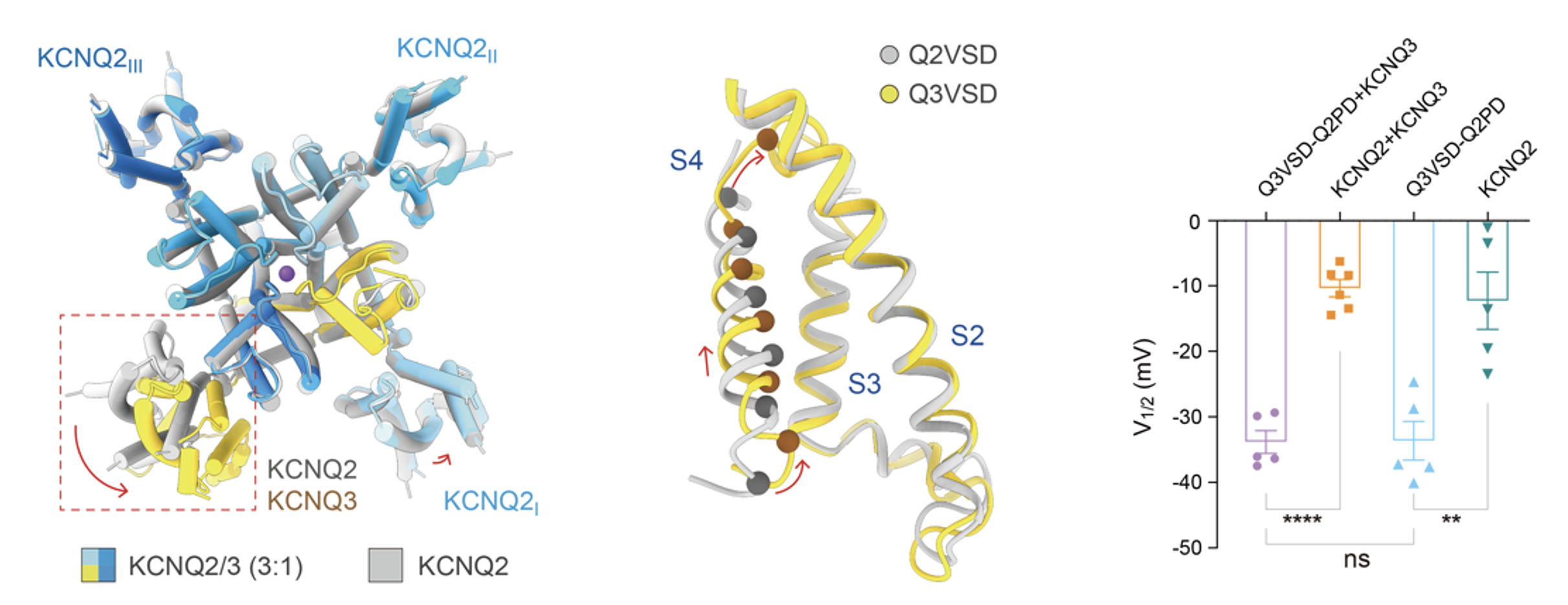
Fig. 3 KCNQ3 decreases the voltage sensitivity of the M-channel
In terms of drug mechanisms, the research team also elucidated the mechanisms of action of two representative M-channel modulators. The ICA-110381 molecule selectively binds to all VSDs of KCNQ2, a property that provides strong corroborating evidence for the distinct subunit identification (Figure 4).
Another compound of great interest, XEN1101, which has entered Phase III clinical trials, binds to a conserved fenestration”(window) site on the side of the pore domain (PD). It acts synergistically with PIP2 to facilitate the gradual opening of the channel in a stepwise activation mode (Figure 5).
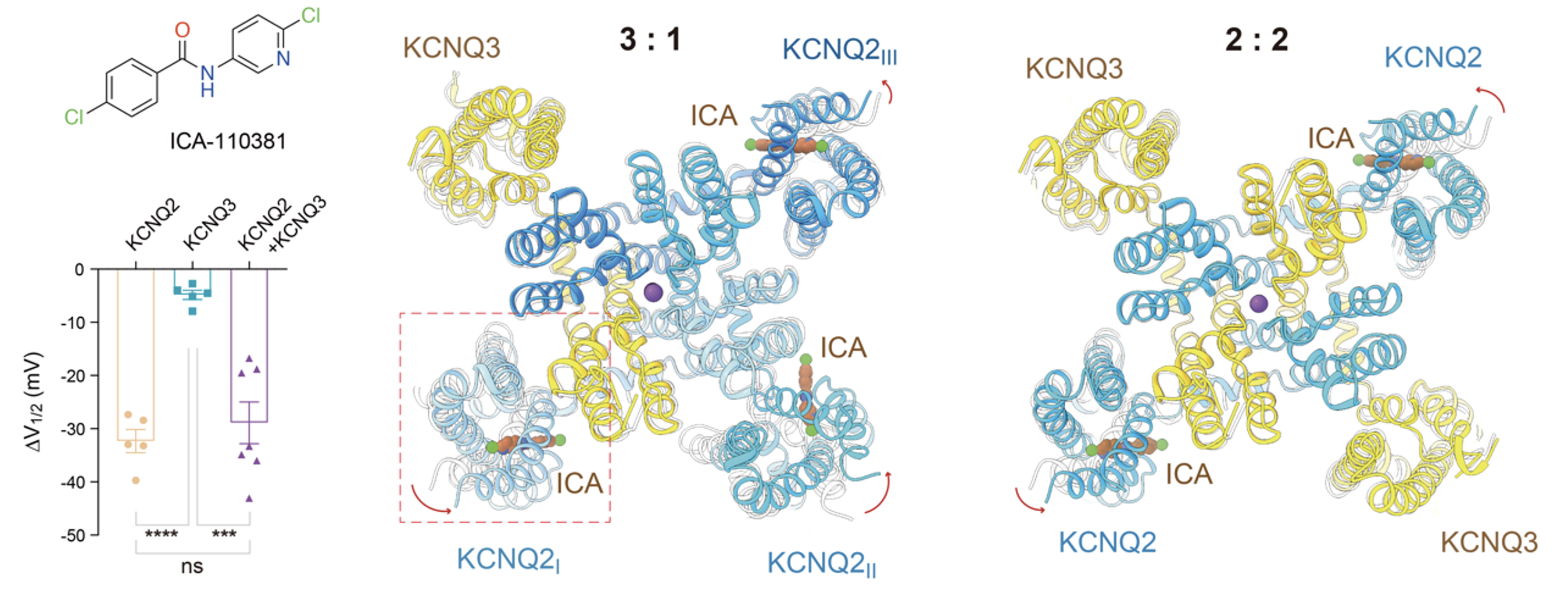
Fig. 4 ICA-110381 selectively targets the voltage-sensing domain of the KCNQ2 subunit
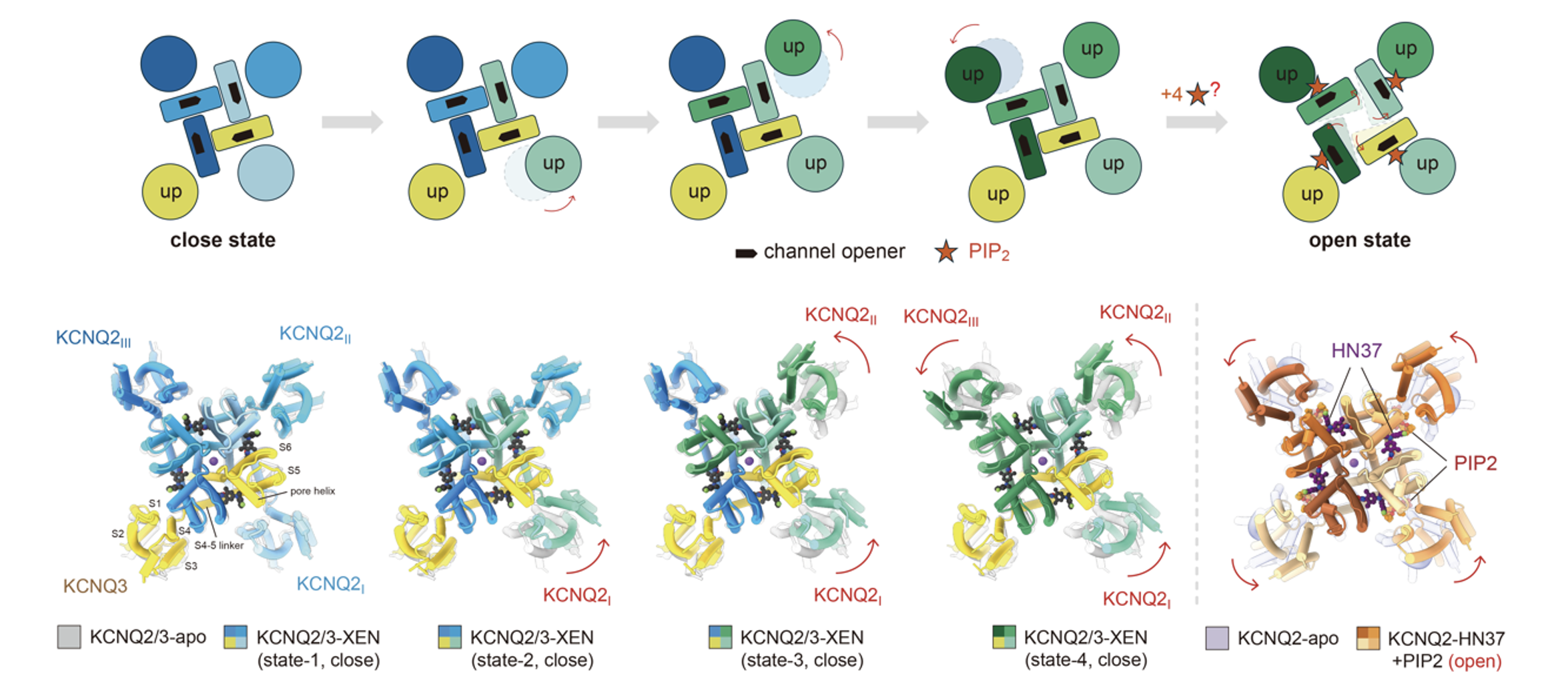
Fig. 5 Stepwise activation mode of the heteromeric M-channel synergistically modulated by XEN1101 and PIP2
This study not only fills a critical gap in the structural biology research of heteromerically assembled M-channels but, more importantly, the subunit-specific drug binding sites and the heteromeric channel stepwise activation mode revealed herein provide an accurate structural template for the development of next-generation antiepileptic drugs with high selectivity, high activity, and low side effects. This is of great significance for the treatment of neurological disorders such as developmental and epileptic encephalopathies.
Jian Huang and Xiao Fan, Junior Principal Investigators at SMART, are the co-corresponding authors of this paper. Fangzhou Lu, a Ph.D. student from the joint program of SMART and Westlake University, and Xiaoshuang Huang, an Associate Investigator at SMART, Guanxing Cai, an engineer at the Electrophysiology Core Facility of SMART, are the co-first authors. Yuzhen Xie, a research assistant in Jian Huang's lab, Ph.D. students from the SMART program, Xianglong Shen, Pei Huang and Feifan Yu, made significant contributions to the research.. This study was strongly supported by the Biological Structure Analysis Center and the Bio-Medical Data Center of SMART, and received funding from SMART and the National Natural Science Foundation of China.
The LTS Preprint Server (LTSpreprints.org) is a scientist-led, open-science platform established by the Shenzhen Medical Academy of Research and Translation (SMART) in collaboration with Tsinghua University, Westlake University, the Shenzhen Bay Laboratory and other leading universities and research institutions.
It also enjoys strong support from the Open Life Science Alliance (openlifesci-alliance.org). Langtaosha Preprint Server provides life sciences researchers worldwide with an efficient, reliable, and open environment for academic communication. The platform facilitates the rapid dissemination of research findings prior to formal peer review, encouraging real-time discussion and fostering broader scientific collaboration.
The simultaneous release of these three studies on the LTS Preprint Server forms a highly complementary and mutually corroborating systematic chain of evidence: from the principles of channel assembly and the remodeling of energy landscapes to the structural explanations of small-molecule regulation and disease-associated mutations, completely reshaping our understanding of M-channels. The rapid posting and sharing through the preprint platform profoundly reflect the core value of open science: shortens the dissemination and communication cycle, allowing researchers in the same field to follow up and cross-validate in a timely manner, thus avoiding redundant effort. Moreover, the “back-to-back” studies mutually reinforce each other to systematically resolve long-disputed questions on M-channel assembly and gating logic. Ultimately, these synergistic breakthroughs provide an atomic-resolution structural blueprint for precise drug design targeting neuroexcitatory diseases such as epilepsy and autism, paving the way for next-generation targeted therapies. This case powerfully demonstrates the crucial role of preprint server in accelerating scientific discovery, promoting cross-team collaboration, and reducing innovation costs. It also provides an instructive open science paradigm for the efficient connection between basic research and clinical translation in the future.
Original link: https://langtaosha.org.cn/index.php/lts/en/preprint/view/59
References:
1. Wulff, H., Castle, N.A. & Pardo, L.A. Voltage-gated potassium channels as therapeutic targets. Nat Rev Drug Discov 8, 982-1001 (2009).
2. Huang, J., Pan, X. & Yan, N. Structural biology and molecular pharmacology of voltage-gated ion channels. Nat Rev Mol Cell Biol 25, 904-925 (2024).
Translation: Yang Shen
Proofreading: Fangzhou Lu
Guo Chen
Subscription successful! Thank you for following SMART.